This guidance document is developed to support UT System Policy, RE0401 Research Misconduct, (henceforth known as the Policy) and to ensure that UT remains compliant with appropriate federal and funding agency requirements applicable to research misconduct. Refer to the Policy for contact information and applicable definitions.
General Information and Introduction
Research Misconduct proceedings are conducted in accordance with these Procedures. A UT campus or institute may depart from these Procedures if necessary to comply with a Research Sponsor Requirement. The Research Integrity Officer (RIO), in consultation with the Involved Dean, the chair of an Inquiry (if convened) or Investigation Committee, Faculty Chairperson, Institutional Deciding Officer (IDO), and/or Chancellor and Office of General Counsel, as appropriate, will decide all procedural issues not addressed by these Procedures, and may supplement these Procedures when necessary to further the Policy or enhance the fairness and efficacy of these Procedures. Appendix A provides a high-level overview of the process for reviewing an allegation of Research Misconduct.
In the event of an allegation against an institutional member (any UT faculty, staff or student) who may also be answerable to a counterpart academic institution or government funding source with respect to the allegation, the RIO may notify the counterpart’s chief academic officer or RIO of the allegation and determine whether to defer action under these Procedures pending assessment, inquiry or investigation of the allegation by the counterpart. If the allegation involves Public Health Service (PHS) funded research, then one institution must be designated as the “Lead Institution.” The Lead Institution should obtain the research records from the other relevant institutions.
During proceedings under these Procedures, at the Respondent’s sole expense, a Respondent may consult an advisor of the Respondent’s choosing and include the advisor at any event under these Procedures to which the Respondent is invited. The Respondent’s advisor is bound to the confidentiality provisions of the Policy. The Respondent’s advisor has no right to participate as an advocate or witness examiner. The RIO, in consultation with committee members, may give courtesy permission for an advisor to assist a Respondent’s presentation of information in proceedings under these Procedures.
The evidentiary standard for an Investigation Committee to find Research Misconduct is a Preponderance of the Evidence considered by the Investigation Committee.
A Respondent will be afforded reasonable prior notice and a reasonable opportunity to be heard by an Investigation Committee, and to present relevant and probative evidence to an Investigation Committee.
Conflicts of Interest
Individuals with a personal, professional or financial interest for or against a Respondent are expected to disclose the interest and offer to recuse themselves from any role in the administration of the Policy. At each stage of these Procedures, conflicts of interest will be solicited from nominated committee members (who will be replaced as appropriate if relevant conflicts emerge), and the Respondent will be given the opportunity to identify to the RIO, within five calendar days, any conflict of interest that the Respondent believes would prevent a nominated committee member or other individual tasked under the Policy from providing fair and unbiased service, describing each perceived conflict in detail.
If the Respondent identifies a conflict of interest involving the RIO and the IDO confirms this conflict of interest, the IDO will appoint another individual to serve as RIO with respect to the allegations against the Respondent. If the Respondent identifies a conflict of interest with another person tasked with administrative responsibilities under this policy and the IDO confirms that conflict of interest, the IDO will appoint an appropriate replacement for that person.
If the Respondent identifies a conflict of interest involving a nominated committee member and the RIO confirms this conflict, the IDO will nominate a substitute committee member. The IDO will receive guidance from the RIO and consult with the Involved Dean as needed for the nomination.
Reports of Research Misconduct
A person who alleges Research Misconduct by a UT institutional member must report the allegation to the RIO. Any allegation of Research Misconduct reported to any other University official, or through any other reporting mechanism, will be forwarded to the RIO. The RIO will forward a copy of each received allegation of Research Misconduct to the IDO.
Admission of Research Misconduct
If a UT institutional member admits to an instance of Research Misconduct, the RIO will submit the admission to the IDO, Provost, Involved Dean, Faculty Chairperson and the Research Sponsor, if any, with a copy to the admitting person. The Provost and/or IDO will determine and impose disciplinary sanctions in accordance with Section XI of these procedures.
In the event that a UT institutional member admits to conduct but disputes whether the admitted conduct is Research Misconduct, or admits to Research Misconduct for fewer than all pending allegations against them, the RIO will conduct an Initial Assessment of the allegations that are not admitted to be Research Misconduct, and proceed accordingly.
Initial Assessment
The purpose of the initial assessment is to determine whether an allegation warrants an inquiry. An assessment is intended to be a review of readily accessible information relevant to the allegation.
Upon receiving an allegation of research misconduct, the RIO or another designated institutional official will promptly determine whether the allegation (a) falls within the definition of research misconduct, (b) falls under the applicability criteria of any applicable federal, state or local regulation or under any funding agency policy, and (c) is credible and specific enough to identify and sequester potential evidence. At the assessment phase, the RIO need not interview the Complainant, Respondent, or other witnesses, or gather data, except as is deemed necessary to complete the assessment. The assessment period should be brief, preferably concluded within two (2) business weeks when possible. In conducting the assessment, the RIO has the discretion to request additional information from the Complainant or consult with experts in the discipline to aid in evaluating the conduct in question.
If the RIO or another institutional official determines that the allegation meets these three criteria, they will promptly: (a) document the assessment and (b) initiate an inquiry and sequester all research records and other evidence. The RIO or other institutional official must document the assessment and retain the assessment documentation securely for a minimum of seven years (depending on state requirements for records retention) after completion of the misconduct proceedings. If the RIO or another institutional official determines that the alleged misconduct does not meet the criteria to proceed to an inquiry, they will write sufficiently detailed documentation to permit a later review by any federal agency or entity as required by law of why UT did not proceed to an inquiry and securely retain this documentation for seven years.
For limited cases, the RIO may proceed directly to an investigation, skipping an inquiry, upon consultation with the Chief Research Officer (CRO) if not the ID), IDO, and Legal Counsel. Limited cases are defined as follows: minimal fact-finding substantiates the allegation; the allegation is referred from a sponsoring entity, including a federal oversight office; a respondent has admitted to some or all of the alleged conduct; OR the allegation has been substantiated by an ongoing or completed audit or other preliminary fact-finding process.
If the three criteria listed above are not met, the RIO should consult with the IDO and/or CRO and University Counsel to determine whether further action is appropriate. Such action may include further review of the allegation under this policy or referral to other institutional compliance officials. When a report of possible research misconduct also contains an allegation of other inappropriate conduct, the allegations will be severed and referred to other offices as is appropriate.
Anonymous Allegations
An allegation reported by an entity, even if no individual complainant is identified, will be handled in the same way as any other allegation. That is, when the allegation is adequately specific and credible to permit meaningful review and is otherwise within the scope of the Policy, the RIO will apply the same standards and criteria for review of all allegations.
Notice of Allegation(s) to Administrators or Oversight Officials
If the RIO determines that an Allegation merits further review under this policy, the RIO (in consultation with University Counsel and the IDO) will notify appropriate University officials. Such notification may include the department head/chair, director, and/or dean, as well as the Chief Academic Officer. The RIO, IDO, and/or CRO may consult with other University officials as needed during the Research Misconduct Proceeding. Because each Research Misconduct Proceeding involves competing concerns, notice to key officials may be determined on a case-by-case basis.
Identifying External Funding and Reporting Obligations
As part of the Assessment, the RIO should identify any external funding that may trigger obligations to federal agencies or other sponsors. The RIO should consult with University Counsel to determine what reporting obligations may be triggered to sponsors when a report of possible Research Misconduct is received. For example, PHS funding is “involved” when a proposal seeking PHS funds is submitted, regardless of whether the proposal is ever reviewed substantively or ultimately funded.
Possible Outcomes of the Assessment
The Assessment is complete when the RIO notifies the Respondent of one or more of these actions:
- Dismissal of the Allegation: The RIO determines that the concern or Allegation (if true) does not meet the definition of Research Misconduct as defined in this policy. The Allegation is dismissed, and the Research Misconduct Proceeding is closed. This includes situations where the RIO concludes that there is not sufficient evidence or information to permit meaningful evaluation of an Allegation.
- Internal Referral: The RIO determines that the conduct reported does not meet the definition of Research Misconduct but merits review by another institutional office or official (e.g., IRB, IACUC, Export Control, or referral to the department head or dean).
- External Referral: The RIO determines that the conduct reported involves a person who is not affiliated with the University such that resolution of the Allegation requires referral to an external entity or institution. Such an external entity includes (but is not limited to) a sponsoring entity, government office or agency, law enforcement, publisher, or other academic institution.
- (a) falls within the definition of research misconduct, (b) falls under the applicability criteria of any applicable federal, state or local regulation or under any funding agency policy, and (c) is credible and specific enough to identify and sequester potential evidence.
- Investigation: In rare situations, the RIO may determine that initial fact-finding conducted through some other internal or external review process indicates that the Allegation may have substance, in which case the RIO may proceed directly to an Investigation.
Written Report of the Assessment
To finalize the Assessment, the RIO will prepare a brief written report containing the following information:
- the name and credentials of each Respondent (including curriculum vitae if possible);
- the source and date of the Allegation;
- a statement of the Allegation with enough specificity to enable the Inquiry committee to conduct a fair and effective review of the conduct reported. The RIO may frame the Allegation more precisely than originally presented by the Complainant. Allegations should be further refined and restated by the RIO or the review committees during the Research Misconduct Proceeding. At a minimum, the Allegation should include:
- the nature of the alleged conduct (Fabrication, Falsification, or Plagiarism);
- the context in which the conduct in question occurred; and
- the specific method of Fabrication, Falsification, or Plagiarism alleged or suspected;
- a description of the evidence collected, reviewed, or sequestered during the Assessment;
- a clear statement of what action should be taken with regard to each Allegation, for each Respondent (e.g., dismissal, referral, Inquiry, or Investigation) and the rationale for each action; and
- any notes or guidance to the committee(s) about issues a committee may need to review.
Written Notice to the Respondent
The timing of Notice to a Respondent may vary depending on the need to obtain the Respondent’s assistance during Sequestration efforts. In any event, the RIO must make a Good Faith effort to notify the Respondent in writing, either before or at the time an Inquiry begins, if the Respondent is known. If no Respondent has been identified, the RIO will notify each potential Respondent that an Inquiry will be undertaken (e.g., each co-author on a questioned publication or each investigator on a questioned grant proposal). Written Notice to the Respondent should include (without limitation):
- a description of the Research being questioned and any external funding involved;
- a statement of the Allegation;
- the definition of Research Misconduct and a copy of (or reference to) this policy; and
- reference to (1) the prohibition against Retaliation, (2) confidentiality requirements, and (3) the Respondent’s duty to cooperate with the Research Misconduct Proceeding.
Appointing an Inquiry or Investigation Committee
An Inquiry and an Investigation differ in both purpose and scope. Under this policy, Allegations of Research Misconduct are reviewed by one or more qualified scholars with the qualifications detailed below.
Appropriate Committee Structure – Specific to Each Proceeding
Whether charged to conduct an Inquiry or Investigation, the process for appointing the review committee is fundamentally the same. The RIO, in consultation with the campus Chief Research Officer and University Counsel, has broad discretion in setting up a review committee most appropriate to the circumstances of a particular Allegation.
|
INQUIRY COMMITTEE MEMBERSHIP (IF CONVENED) |
INVESTIGATION COMMITTEE MEMBERSHIP |
|
The RIO appoints an Inquiry committee consisting of at least one person who meets the requirements for committee membership outlined below. |
The RIO appoints an Investigation committee consisting of at least three people who meet the requirements for committee membership outlined below. |
Identifying Committee Nominees
The RIO may solicit recommendations for committee membership from any source, and will ensure that anyone consulted in that process is aware of the University’s confidentiality obligations. The RIO will contact each potential member of the committee to determine whether he/she is available to serve, to confirm that he/she has the required expertise, and to identify any potential Conflict of Interest.
Committee members will be selected based on their expertise and integrity, and:
- Must have appropriate expertise to evaluate the evidence and issues related to the Allegation, interview the principals and key witnesses, and otherwise be capable of conducting a full and fair review;
- Must not have any unresolved personal, professional, or financial Conflicts of Interest with any Respondent, Complainant, or anyone expected to be a critical witness;
- May be drawn from the same campus or from other UT campuses, and may include members from outside the University if adequate expertise and independence is not available within the University, or if the RIO determines that outside perspective is necessary to conduct an effective Inquiry;
- May have served on a previous committee in this (or another) Research Misconduct Proceeding or in any other capacity in any previous proceeding, so long as he/she otherwise meets the requirements for committee membership.
Prohibited Conflicts of Interest
Unresolved Conflicts of Interest among key participants in a Research Misconduct Proceeding are prohibited. As defined in Section 2, a Conflict of Interest for the purpose of this policy means a past or continuing personal, professional, or financial relationship with another participant in the Research Misconduct Proceeding which creates an unmanageable (as determined by the RIO) bias for or against any Respondent, or which the participant him/herself personally believes renders him/her unable to participate in a manner that is thorough, competent, objective, and fair.46
Standing alone, service in some other role during a Research Misconduct Proceeding (the same proceeding or a different one) does not constitute a Conflict of Interest. Specifically, service on an Inquiry committee in the same Research Misconduct Proceeding does not create a Conflict of Interest relevant to that person’s service on a later Investigation committee in the same (or other) Research Misconduct Proceeding.
If a Respondent asserts that the RIO has a prohibited Conflict of Interest, the purported conflict will be reviewed by the Chief Research Officer in consultation with other officials as the Chief Research Officer may find appropriate. The Chief Research Officer’s decision regarding the RIO’s eligibility to serve in any particular case will be final. If the RIO is unable serve in a particular case because of a Conflict of Interest or any other reason, the Chief Research Officer shall appoint an interim RIO as he/she deems appropriate. If a Respondent asserts that the Chief Research Officer has a Conflict of Interest, the Chief Academic Officer shall review the purported conflict and will act in place of the Chief Research Officer as he/she deems appropriate.
Respondent’s Opportunity to Challenge Committee Nominations
The RIO will provide the Respondent an opportunity to object in writing to the appointment of any person to the committee on the basis that the person has an unresolved Conflict of Interest. To challenge the appointment of any nominee, the Respondent must identify and explain in writing the perceived Conflict of Interest within 5 calendar days of RIO’s transmittal of the nomination. If the Respondent provides a timely written objection to a nominee, the RIO will review the purported Conflict of Interest with the nominee and other appropriate officials and will make a decision on the nominee’s eligibility for service on the committee. The RIO’s decision regarding composition of the committee is final.
Finalizing Committee Appointments
Once the membership of the committee is finalized, the RIO shall select a chair. Letters appointing members of the committee (but not identifying the Respondent by name) will be provided to each committee member, with a copy provided to his/her department head unless otherwise requested. Service on a Research Misconduct committee is challenging and time consuming; such service should be recognized and rewarded as may be appropriate under relevant departmental, college, or campus evaluation criteria.
RIO’s First Meeting with Committee
The RIO and University Counsel will be present or available throughout the proceeding to advise and support the committee. At the committee’s first meeting, the RIO should:
- Review the charge;
- Summarize procedures for conducting the Inquiry/Investigation, and identify available resources;
- Describe Sequestration efforts to date and any security measures in place;
- Lead an initial discussion of the nature of the Allegation(s);
- Identify documents to be requested, Sequestration to be undertaken, or other support the RIO can provide;
- Guide the committee in organizing and planning the Inquiry/Investigation process, including establishing a schedule of meetings, and the eventual need to prepare a written report.
Inquiry
An inquiry is warranted if the allegation (a) falls within the definition of research misconduct, (b) falls under the applicability criteria of any applicable federal, state or local regulation or under any funding agency policy, and (c) is credible and specific enough to identify and sequester potential evidence. An inquiry’s purpose is to conduct an initial review of the evidence to determine whether an allegation warrants an investigation. An inquiry does not require a full review of all related evidence. Each UT campus and institute will complete the inquiry within 90 days of initiating it unless circumstances warrant a longer period, in which it will sufficiently document the reasons for exceeding the time limit in the inquiry report. Additional resources for an inquiry can be found in Appendix B.
Sequestering Evidence and Notifying the Respondent
Before or at the time of notifying the respondent(s), UT will obtain the original or substantially equivalent copies of all research records and other evidence that are pertinent to the proceeding, inventory these materials, sequester the materials in a secure manner, and retain them for seven years. The institution has a duty to obtain, inventory, and securely sequester evidence that extends to whenever additional items become known or relevant to the inquiry or investigation.
At the time of or before beginning the inquiry, UT will make a good-faith effort to notify the presumed respondent(s), in writing, that an allegation(s) of research misconduct has been raised against them, the relevant research records have been sequestered, and an inquiry will be conducted to decide whether to proceed with an investigation. If additional allegations are raised, the institution will notify the respondent(s) in writing. When appropriate, the institution will give the respondent(s) copies of, or reasonable supervised access to, the sequestered materials.
If additional respondents are identified, UT will provide written notification to the new respondent(s). All additional respondents will be given the same rights and opportunities as the initial respondent. Only allegations specific to a particular respondent will be included in the notification to that respondent.
If a respondent is not available to meet with the RIO and conduct the initial sequestration, the RIO may proceed in the Respondent’s absence. In that situation, the RIO will make efforts to communicate with the respondent once sequestration is in process, in order to focus the process as much as possible. In the event that the respondent refuses to provide access to evidence identified by the RIO as requiring sequestration, the RIO has the obligation and authority to enter any University-owned or -controlled property (with law enforcement assistance if necessary) to retrieve University property.
Convening the Committee and Ensuring Neutrality
UT will ensure that all inquiry committee members, if convened, understand their commission, keep the identities of respondents, complainants, and witnesses confidential, and conduct the research misconduct proceedings in compliance with any applicable federal or funding agency regulation and requirement. In lieu of a committee, the institution may task the RIO or another designated institutional official to conduct the inquiry, provided this person utilizes subject matter experts as needed to assist in the inquiry.
Determining Whether an Investigation Is Warranted
The inquiry committee, RIO, or other designated institutional official will conduct a preliminary review of the evidence. In the process of fact-finding, the inquiry committee may interview the respondent and/or witnesses. An investigation is warranted if (a) there is a reasonable basis for concluding that the allegation falls within the definition of research misconduct as defined in the policy and meets any other applicable requirements of a federal regulation or law and funding agency requirement; and (b) preliminary information-gathering and fact-finding from the inquiry indicates that the allegation may have substance.
The inquiry committee will not determine if research misconduct occurred, nor assess whether the alleged misconduct was intentional, knowing, or reckless; such a determination is not made until the case proceeds to an investigation.
Documenting the Inquiry
At the conclusion of the inquiry, regardless of whether an investigation is warranted, the inquiry committee, RIO, or other designated institutional official will prepare a written inquiry report. The contents of a complete inquiry report will include:
- The names, professional aliases, and positions of the respondent and complainant(s).
- A description of the allegation(s) of research misconduct.
- Details about the source of funding, including any grant numbers, grant applications, contracts, and publications listing PHS support.
- The composition of the inquiry committee, if used, including name(s), position(s), and subject matter expertise.
- An inventory of sequestered research records and other evidence and description of how sequestration was conducted.
- Transcripts of interviews, if transcribed.
- Inquiry timeline and procedural history.
- Any scientific or forensic analyses conducted.
- The basis for recommending that the allegation(s) warrant an investigation.
- The basis on which any allegation(s) do not merit further investigation.
- Any comments on the inquiry report by the respondent or the complainant(s).
- Any institutional actions implemented, including internal communications or external communications with journals or funding agencies.
- Documentation of potential evidence of honest error or difference of opinion.
Completing the Inquiry
The respondent will be given a copy of the draft inquiry report for review and comment by the RIO. The institution may, but is not required to, provide relevant portions of the report to a complainant for comment.
The respondent will be notified of the inquiry’s final outcome and provide the respondent with copies of the final inquiry report, the applicable federal or funding agency regulation, a copy of RE0402 Research Misconduct Policy, this guidance document, and any additional campus or institute specific procedures. The RIO may, but is not required to, notify a complainant whether the inquiry found that an investigation is warranted. If the notice is provided to one complainant in a case, it must provide notice, to the extent possible, to all complainants in the case.
If an Investigation Is Not Warranted:
If the inquiry committee, RIO, or other designated institutional official determines that an investigation is not warranted, the UT campus or institute will keep sufficiently detailed documentation to permit a later review by an outside entity as required by law or regulation (e.g., ORI, NSF) of why the institution did not proceed to an investigation, store these records in a secure manner for at least seven years after the termination of the inquiry, and provide them upon request.
If an Investigation is Warranted:
If the inquiry committee, RIO, or other designated institutional official determines that an investigation is warranted, UT must: (a) within a reasonable amount of time after this decision, provide written notice to the respondent(s) of the decision to conduct an investigation of the alleged misconduct, including any allegations of research misconduct not addressed during the inquiry; and (b) within 30 days of determining that an investigation is warranted, provide external agencies or entities with a copy of the inquiry report, as required by their regulations.
On a case-by-case basis, UT may choose to notify the complainant that there will be an investigation of the alleged misconduct but is required to take the same notification action for all complainants in cases where there is more than one complainant.
The purpose of an investigation is to formally develop a factual record, pursue leads, examine the record, and recommend finding(s) to the IDO, who will make the final decision, based on a preponderance of evidence, on each allegation and any institutional actions. As part of its investigation, the institution will pursue diligently all significant issues and relevant leads, including any evidence of additional instances of possible research misconduct, and continue the investigation to completion. Within 30 days after deciding an investigation is warranted, UT will notify any federal agency or entity of the decision to investigate and begin the investigation, as required by regulation. Additional resources for an inquiry can be found in Appendix B.
Notifying the Respondent and Sequestering Evidence
UT will notify the respondent(s) of the allegation(s) within 30 days of determining that an investigation is warranted and before the investigation begins. If any additional respondent(s) are identified during the investigation, those individuals will be notified by the RIO of the allegation(s) and provide them with an opportunity to respond. If UT identifies additional respondents during the investigation, it may choose to either conduct a separate inquiry or add the new respondent(s) to the ongoing investigation. UT will obtain the original or substantially equivalent copies of all research records and other evidence, inventory these materials, sequester them in a secure manner, and retain them for seven years after its proceeding or any external agency or entity proceeding (e.g., HHS), whichever is later.
Convening an Investigation Committee
After vetting investigation committee members for conflicts of interest and appropriate scientific expertise, UT will convene the committee and ensure that the members understand their responsibility to conduct the research misconduct proceedings in compliance with these procedures or any federal agency or entity regulation. The investigation committee will conduct interviews, pursue leads, and examine all research records and other evidence relevant to reaching a decision on the merits of the allegation(s). UT will use diligent efforts to ensure that the investigation is thorough, sufficiently documented, and impartial and unbiased to the maximum extent practicable. UT will notify the respondent in writing of any additional allegations raised against them during the investigation.
Conducting Interviews
The assembled investigation committee will interview each respondent, complainant(s), and any other available person who has been reasonably identified as having information regarding any relevant aspects of the investigation, including witnesses identified by the respondent. All relevant exhibits will be numbered and reference to any exhibits shown to the interviewee during the interview by that number. The interviews will be recorded and transcribed interviews during the investigation and the transcripts made available to the interviewee for correction. The transcript(s) will be included with any corrections and exhibits in the institutional record of the investigation. The respondent will not be present during the witnesses’ interviews, but the institution will provide the respondent with a transcript of each interview, with redactions as appropriate to maintain confidentiality.
Documenting the Investigation
Each UT campus and institute will complete all aspects of the investigation within 180 days. UT will conduct the investigation, prepare the draft investigation report for each respondent, and provide the opportunity for respondents to comment. The institution will document the IDO’s final decision and transmit the institutional record (including the final investigation report and IDO’s decision) to external federal agencies and entities as required, if applicable. If the investigation takes more than 180 days to complete, and the investigation involves a federal agency or entity (e.g., ORI), UT will ask, in writing, for an extension and document the reasons for exceeding the 180-day period in the investigation report.
The investigation report for each respondent will include:
- Description of the nature of the allegation(s) of research misconduct, including any additional allegation(s) addressed during the research misconduct proceeding.
- Description and documentation of any funding support, including any grant numbers, grant applications, contracts, and publications listing sponsor support. This documentation includes known applications or proposals for support that the respondent has pending Federal agencies.
- Description of the specific allegation(s) of research misconduct for consideration in the investigation of the respondent.
- Composition of investigation committee, including name(s), position(s), and subject matter expertise.
- Inventory of sequestered research records and other evidence, except records the institution did not consider or rely on. This inventory will include manuscripts and funding proposals that were considered or relied on during the investigation. The inventory will also include a description of how any sequestration was conducted during the investigation.
- Transcripts of all interviews conducted.
- Identification of the specific published papers, manuscripts submitted but not accepted for publication (including online publication), funding applications, progress reports, presentations, posters, or other research records that contain the allegedly falsified, fabricated, or plagiarized material.
- Any scientific or forensic analyses conducted.
- A copy of these policies and procedures.
- Any comments made by the respondent and complainant(s) on the draft investigation report and the committee’s consideration of those comments.
- A statement for each separate allegation of whether the committee recommends a finding of research misconduct.
If the committee recommends a finding of research misconduct for an allegation, the investigation report will present a finding for each allegation. These findings will (a) identify the individual(s) who committed the research misconduct; (b) indicate whether the misconduct was falsification, fabrication, and/or plagiarism; (c) indicate whether the misconduct was committed intentionally, knowingly, or recklessly; (d) identify any significant departure from the accepted practices of the relevant research community and that the allegation was proven by a preponderance of the evidence; (e) summarize the facts and analysis supporting the conclusion and consider the merits of any explanation by the respondent; (f) identify the specific sponsor support; and (g) state whether any publications need correction or retraction.
If the investigation committee does not recommend a finding of research misconduct for an allegation, the investigation report will provide a detailed rationale for its conclusion.
The investigation committee should also provide a list of any current support or known applications or proposals for support that the respondent has pending with PHS and non-PHS Federal agencies.
Completing the Investigation
UT will give the respondent a copy of the draft investigation report and, concurrently, a copy of, or supervised access to, the research records and other evidence that the investigation committee considered or relied on. The respondent will submit any comments on the draft report to the institution within 30 days of receiving the draft investigation report. If UT chooses to share a copy of the draft investigation report or relevant portions of it with the complainant(s) for comment, the complainant’s comments will be submitted within 30 days of the date on which they received the report. UT will add any comments received to the investigation report. Additional resources are provided in Appendix D.
IDO Review of the Investigation Report
The IDO will review the investigation report and make a final written determination of whether the institution found research misconduct and, if so, who committed the misconduct. In this statement, the IDO will include a description of relevant institutional actions taken or to be taken. Additional resources are provided in Appendix C.
Creating and Transmitting the Institutional Record
After the IDO has made a final determination of research misconduct findings, UT will add the IDO’s written decision to the investigation report and organize the institutional record in a logical manner.
The institutional record consists of the records that were compiled or generated during the research misconduct proceeding, except records the institution did not rely on. These records include documentation of the assessment, a single index listing all research records and evidence, the inquiry report and investigation report, and all records considered or relied on during the investigation. The institutional record also includes the IDO’s final decision and any information the respondent provided to the institution. The institutional record must also include a general description of the records that were sequestered but not considered or relied on.
If the respondent filed an appeal, the complete record of any institutional appeal also becomes part of the institutional record. UT will wait until the appeal is concluded to transmit the institutional record to any required external federal agency or entity (e.g., ORI). After the IDO has made a final written determination, and any institutional appeal is complete, UT must transmit the institutional record to all applicable UT leadership (UT President, General Counsel, AVP for Research, Chancelor, Provost, Vice Chancelor for Research, Involved Dean and external federal agency or entity as applicable).
Other Procedures and Special Circumstances
Multiple Institutions and Multiple Respondents
If the alleged research misconduct involves multiple institutions, UT may work closely with the other affected institutions to determine whether a joint research misconduct proceeding will be conducted. If so, the cooperating institutions will choose an institution to serve as the lead institution. In a joint research misconduct proceeding, the lead institution will obtain research records and other evidence pertinent to the proceeding, including witness testimony, from the other relevant institutions. By mutual agreement, the joint research misconduct proceeding may include committee members from the institutions involved. The determination of whether further inquiry and/or investigation is warranted, whether research misconduct occurred, and the institutional actions to be taken may be made by the institutions jointly or tasked to the lead institution.
If the alleged research misconduct involves multiple respondents, the UT campus or institute involved may either conduct a separate inquiry for each new respondent or add them to the ongoing proceedings. The institution must give additional respondent(s) notice of and an opportunity to respond to the allegations.
Respondent Admissions
UT will promptly notify external federal agencies and/or entities (e.g., ORI) in advance if at any point during the proceedings (including the assessment, inquiry, investigation, or appeal stage) it plans to close a research misconduct case because the respondent has admitted to committing research misconduct or a settlement with the respondent has been reached.
In the case of PHS research misconduct, if the respondent admits to research misconduct, the institution will not close the case until providing ORI with the respondent’s signed, written admission. The admission must state the specific fabrication, falsification, or plagiarism that occurred, which research records were affected, and that it constituted a significant departure from accepted practices of the relevant research community. UT must not close the case until giving ORI a written statement confirming the respondent’s culpability and explaining how the institution determined that the respondent’s admission fully addresses the scope of the misconduct.
Other Special Circumstances
At any time during the misconduct proceedings, UT will immediately notify applicable UT leadership (UT President, General Counsel, AVP for Research, Chancelor, Provost, Vice Chancelor for Research, Involved Dean and external federal agency or entity as applicable) if any of the following circumstances arise:
- Health or safety of the public is at risk, including an immediate need to protect human or animal subjects.
- Funding sponsor resources or interests are threatened.
- Research activities should be suspended.
- There is reasonable indication of possible violations of civil or criminal law.
- Federal action is required to protect the interests of those involved in the research misconduct proceeding.
- Funding sponsor may need to take appropriate steps to safeguard evidence and protect the rights of those involved.
UT will maintain the institutional record and all sequestered evidence, including physical objects (regardless of whether the evidence is part of the institutional record), in a secure manner for a minimum seven years after the completion of the proceeding or the completion of any external federal agency or entity proceeding (e.g., HHS) or any state records retention requirements, whichever is later, unless custody has been transferred to HHS.
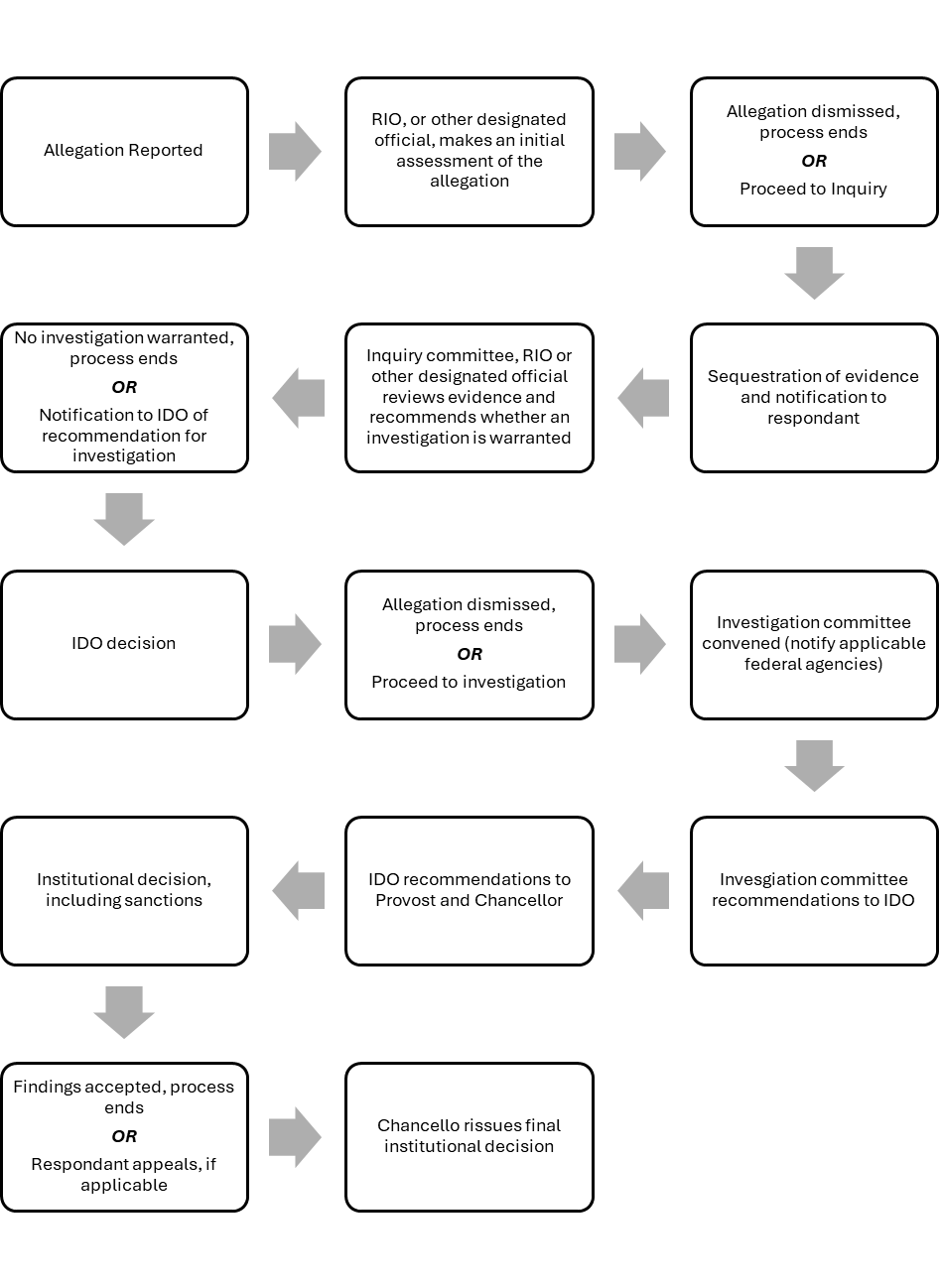
Appendix A: Process for Reviewing Alleged Research Misconduct
Appendix B: Additional Information relating to Conducting an Inquiry or Investigation
|
WHEN TO CONDUCT AN INVESTIGATION | |
|
An Inquiry or other Research Misconduct Proceeding will be initiated after the RIO has conducted an Assessment and determined that an Inquiry is appropriate, that is whether the Allegation (if true)
|
An Investigation will be initiated when the IDO concludes (after reviewing the report of an Inquiry committee or other preliminary review of the Allegation):
|
|
BEFORE AN INVESTIGATION | |
|
The RIO must make a good faith effort to notify the Respondent in writing, either before or at the time an Inquiry begins, if the Respondent is known. If not, the RIO will notify each potential Respondent that an Inquiry will be undertaken (e.g., each co-author on a questioned publication or each investigator on a questioned grant proposal). Written Notice to the Respondent should include (without limitation):
|
When the IDO determines that Investigation is warranted:
|
|
INVESTIGATION TIMEFRAME | |
|
The University attempts to complete an Inquiry within 90 days from the date the committee is charged. The committee should attempt to complete its review during the first 30-45 days, to allow time to draft a report and obtain review by the RIO and University Counsel. |
The University attempts to complete an Investigation within 180 days from the date the committee is charged. The committee should attempt to complete its review during the first 90 days, to allow time to draft a report, obtain review by the RIO and University Counsel, provide a draft report to the Respondent (with 10 calendar days to respond in writing), consider the Respondent’s response, and finalize the report. |
If allegations or Respondents are added during the proceeding, the time for completion will normally be extended. All extensions should be documented, including the reasons supporting the extension. The RIO will notify the Respondent of extensions.
The RIO may grant an extension (or request an extension from oversight officials) whenever he/she deems it necessary in the interest of providing a fair process for any Respondent, witness, or other participant in the proceeding OR when it is otherwise in the best interest of the University to do so. Continuation of the case over the summer months is not a valid reason for an extension. Committee members are expected to make themselves available (at least by phone or videoconference) for a series of meetings during the projected time span of the Inquiry/Investigation.
Adding/Revising the Allegation(s) or Adding Respondent(s) – Required Notice
During the Research Misconduct Proceeding additional instances of possible Research Misconduct may be discovered, which may require expanding the scope of the review beyond the Allegation(s) stated in the charge to the committee. Federal regulations and this policy require that the University attempt to discover any similar Research Misconduct that may have occurred by the same Respondent (e.g., in other publications). If the RIO, Inquiry committee, or Investigation committee identifies evidence that creates a reasonable basis to conclude that newly discovered conduct falls within the definition of Research Misconduct, the newly discovered conduct must undergo further review, either as a part of a revised Allegation or as a new Allegation, as determined by the RIO. Federal regulations and this policy require that the University attempt to discover any similar Research Misconduct that may have occurred by the same Respondent.
The RIO will promptly notify a Respondent of any new or revised Allegation or any new Allegation identified during the Inquiry or Investigation, and will provide initial Notice to any new Respondent of the ongoing proceedings. Although the Research Misconduct Proceeding does not begin anew for such a Respondent, his/her fundamental rights are nonetheless preserved: specifically, each Respondent is given written Notice of the Allegation and the opportunity to respond to the Allegation.
Substantive Questions for the Committee’s Consideration
|
INVESTIGATION QUESTION(S) TO BE ANSWERED | |
|
The purpose of the Inquiry is to conduct an initial review of the available evidence to determine whether the Allegation meets the criteria warranting an Investigation. Specifically, the committee is charged to make findings of fact and recommendations as to whether:
|
The purpose of an Investigation is to develop a factual record by exploring the Allegation(s) in detail and examining the evidence in depth, leading to conclusions and recommendations by the committee using the following standard: In the process of proposing, conducting, reviewing or reporting Research, a Respondent has committed Research Misconduct when a Preponderance of the Evidence supports a finding that
Any defense raised by the Respondent (such as Honest Error or difference of opinion) must be established by a Preponderance of the Evidence. The absence of Research Records may be evidence of Research Misconduct when it is established by a Preponderance of the Evidence that the Respondent once had possession of the records but significantly departed from accepted practices of the relevant Research community when he/she Intentionally, Knowingly, or Recklessly destroyed, failed to maintain, or failed to produce such records in a timely manner. |
Key Elements of the Review
|
KEY COMPONENTS OF THE INQUIRY |
KEY COMPONENTS OF THE INVESTIGATION |
|
Data and Materials: The Inquiry committee (through the RIO) requests or collects any documents, data, or other materials which the committee finds necessary to complete its charge. |
Data and Materials: The Investigation committee (through the RIO) requests or collects any documents, data, or other materials which the committee finds necessary to complete its charge. |
|
Interviewing the Respondent: The Inquiry committee should interview each Respondent unless the Respondent:
A Respondent may provide information to the committee in person, by phone or videoconference, and/or in writing, at the Respondent’s discretion. |
Interviewing the Respondent: The Investigation committee must interview each Respondent unless the Respondent:
A Respondent may provide information to the committee in person, by phone or videoconference, and/or in writing, at the Respondent’s discretion. |
|
Interviewing the Complainant: The Inquiry committee will normally interview the Complainant. |
Interviewing the Complainant: The Investigation committee must interview each Complainant unless the Complainant declines to be interviewed or fails to respond to a request for an interview. |
|
Interviewing Other Witnesses: If the Inquiry committee concludes that interviewing additional witnesses is necessary to complete its limited charge, it may do so, but the committee should not conduct exhaustive interviews or analysis. |
Interviewing Other Witnesses: The Investigation committee must interview any other available person who has been reasonably identified as having information regarding any relevant aspect of the Investigation, including witnesses identified by the Respondent. |
|
Recording Interviews: An interview of the Respondent must be recorded or transcribed. All other interviews should be recorded or transcribed but if not, a written summary of the interview must be prepared. The recording, transcript, or summary will be provided to the interviewee for correction and must be included in the record of the Investigation |
Recording Interviews: All interviews must be recorded or transcribed. The recording or transcript will be provided to the interviewee for correction and must be included in the record of the Investigation. |
|
New or Revised Allegations: The Inquiry committee should revise, eliminate, or add Allegations as the evidence suggests, and should add other Respondents as deemed necessary. |
New or Revised Allegations: The Investigation committee must pursue diligently all significant issues and leads discovered that are determined, by the committee or the RIO, to be relevant to the Investigation, including evidence of additional instances of possible Research Misconduct, and must complete a full Investigation unless otherwise advised by the RIO. |
|
Conclusion: The Inquiry committee must recommend whether an Investigation is warranted, but should not make any determination of whether Research Misconduct occurred. |
Conclusion: The Investigation committee must conclude whether Research Misconduct occurred, and if so, who was responsible. |
The committee will document its findings and recommendations in a written report containing at least the elements listed below. The committee must submit a draft of the report to the RIO and University Counsel for review of compliance with this policy and legal sufficiency.
|
INQUIRY REPORT CHECKLIST |
INVESTIGATION REPORT CHECKLIST |
|
1. Name and position of the Respondent: Although a separate report must be prepared for each Respondent, portions of the reports may be similar (e.g., background and factual summaries); | |
|
2. List (with supporting documents attached) of funding sources related to the Research under review, particularly any basis for federal oversight jurisdiction (e.g., PHS support, citation of PHS-funded Research as preliminary data), including any relevant grant or proposal numbers, contracts, or publications; | |
|
3. Name and title of each committee member, attaching a current curriculum vitae for each member; | |
|
4. List of interviews conducted, attaching transcripts or interview summaries; | |
|
5. A copy of the written charge to the committee, with any attachments; | |
|
6. Statement of the specific Allegation(s) reviewed; | |
|
7. Summary of Sequestration undertaken and, if possible, a copy of the evidence log; | |
|
8. Narrative summary of the evidence and records reviewed (with a list of attachments, where appropriate); | |
|
9. For each Allegation of Research Misconduct, an explanation of the basis for the Inquiry Committee’s recommendation that an Investigation is or is not warranted. |
9. For each Allegation of Research Misconduct, a detailed explanation of the Investigation committee’s findings regarding:
|
|
10. Discussion of plausible mitigating explanations provided by Respondent; | |
|
11. Citation information for any publications in need of correction or retraction; and | |
|
12. Impact of any written response to the draft report submitted by the Respondent. | |
Respondent’s Opportunity to Comment on the Committee’s Report
|
RESPONDENT’S REVIEW OF DRAFT INVESTIGATION REPORT | |
|
The RIO must give each Respondent Notice of the Inquiry committee’s findings and recommendations including:
|
The RIO must provide each Respondent with a draft of the Investigation report. The Respondent may submit a written response to the draft report within 10 calendar days of transmittal from the RIO. |
Complainant’s Opportunity to Comment on the Committee’s Report
At the RIO’s discretion, the University may:
- notify a Complainant of the Inquiry committee’s findings; or
- provide the Complainant with a copy of the Inquiry report (in draft or final form) or relevant portions of the report; or
- invite a written response from the Complainant to the findings or the written report (or portions thereof).
If the RIO permits a written response from the Complainant, that response will be reviewed and considered by the committee if it is received within 10 calendar days of the date on which it is provided to the Complainant. If the committee receives and considers a Complainant’s response, it will be included as an attachment to the final report.
After reviewing the findings and recommendations of the committee, together with any further information or recommendations provided by the RIO (normally within 30 days of receiving the report) the IDO will take one or more of the following responses:
- Accept the report in full;
- Return the report to the committee with instructions or questions for further consideration;
- Make alternative findings, including a detailed explanation for doing so; OR
- Reject the report in full, with a written explanation for the rejection.
Appendix C: Additional Information on Making and Reporting a Finding of Research Misconduct
IDO Finding of Research Misconduct Based on an Investigative Report
After reviewing the findings and recommendations of the Investigation committee, together with any further information or recommendations provided by the RIO (normally within 30 days of receiving the report) the IDO will take one or more of the following actions:
-
- Accept the report (in whole or in part);
OR
-
- Make alternate findings by offering a detailed explanation of his/her reasons for doing so;
OR
-
- Return the report to the RIO and Investigation committee for further action. Specifically, the IDO may return the report to the RIO and the committee with a request for further review, additional findings of fact, or clarification.
The IDO’s written decision is provided to the Respondent and to appropriate University officials, and to any oversight officials as required by applicable regulations or contracts.
IDO Finding of Research Misconduct Based on a Fully Acceptable Admission
Except in rare and specific circumstances, all Inquiries and Investigations will be carried through to completion, and all plausible and significant concerns of possible Research Misconduct will be reviewed. The RIO must notify appropriate oversight officials in advance if the University considers closing a case prior to completion of a full Investigation for any reason, including (without limitation) an admission of guilt by the Respondent or resolution of the Allegation through a negotiated agreement.
In a Research Misconduct Proceeding over which federal oversight officials (particularly PHS-ORI and NSF-OIG) have jurisdiction, the University is not permitted to accept an admission of guilt or other settlement arrangement without advance approval from oversight officials. If the RIO finds that it may be appropriate to negotiate a resolution or close a case prior to completion of the Investigation, he/she will first review the issue with the campus or institute Chief Research Officer and University Counsel before taking further action to close the matter or seeking permission from oversight officials to close the Research Misconduct Proceeding before completion of a full Investigation.
Normally, an admission of Research Misconduct is a sufficient basis for proceeding directly to the Investigation stage. However, an admission may not be a sufficient basis for closing a Research Misconduct Proceeding.
An Investigation is usually required to determine (or confirm) the extent of the Research Misconduct or to explore other possible instances of Fabrication, Falsification, or Plagiarism or to consider other possible Respondents who may share in the responsibility for Research Misconduct. Specifically, an admission of Research Misconduct during the early stages of a Research Misconduct Proceeding does not eliminate the need for the RIO to sequester evidence confirming the extent of the Research Misconduct. Federal regulations require that the University preserve all potential evidence of Research Misconduct until the case is concluded by federal oversight officials, including approval of an admission.
An admission of Research Misconduct requires specific admission of all elements of the evidentiary standard and must:
- be made in writing, signed, dated, and witnessed;
- be made freely, without coercion or duress;
- identify the specific instances of Fabrication, Falsification, or Plagiarism;
- explicitly acknowledge that the conduct admitted constitutes Research Misconduct;
- explain the manner in which the Fabrication, Falsification, or Plagiarism was conducted or included in the Research Record; and
- identify any PHS or other external funding involved in the conduct at issue.
If the Research Misconduct Proceeding is closed based on an approved admission, a report summarizing the steps taken in accepting the admission (including documentation of approval of the admission by federal oversight officials) will be sent to the IDO, including any recommendations for Administrative Action or Sanctions.
Appeal Rights after a Finding of Research Misconduct
A decision by the IDO that a Preponderance of the Evidence supports the finding that a Respondent committed a specific act of Research Misconduct is final and unappealable.
In contrast, Administrative Actions or Sanctions imposed in response to a finding of Research Misconduct may be appealed through the normal appeals processes applicable to the Respondent, except that (at the IDO’s discretion) such actions need not be held in abeyance pending appeal. For example, faculty appeals may be taken through the process described in Board policies and the applicable faculty handbook; students may appeal in a manner described in the applicable student handbook; and staff may appeal under applicable HR policies.
Reporting to Oversight Officials
In cases where external funding is involved, the IDO will report a finding of Research Misconduct to the appropriate officials within the granting agency or as required under the contract. The report should include actions proposed or imposed by the University in response to a finding of Research Misconduct. Such actions may include remedial, administrative, or disciplinary action against a Respondent, as well as lab-, department-, or campus-wide training initiatives. Revision of policies may also be recommended to the appropriate University office (e.g., safety policies, IRB approval processes).
Internal referral of a concern (including limited disclosure to a University official regarding the University’s response to a finding of Research Misconduct) is consistent with the confidentiality requirements of the PHS regulations and this policy and procedures.
Appendix D: Closure of a Proceeding, Administrative Actions, and Sanctions
Initial Remedial, Administrative, or Disciplinary Actions
This policy identifies some of the common remedial, administrative, and disciplinary actions the University may take in response to a finding of Research Misconduct; however, no list of possible responses could cover every case. The IDO will consult with University Counsel before initiating any action in response to a finding of Research Misconduct.
-
- For remedial and Administrative Actions, the IDO will advise the Chief Academic Officer and other appropriate academic officials of the actions proposed or imposed by the IDO.
- For disciplinary action, the IDO will consult with academic administrators, HR officials, or student disciplinary officials about possible disciplinary action. After consultation, the IDO may conclude that the disciplinary action should be initiated by a department head, dean, Chief Academic Officer, or other administrator. The procedure for imposing disciplinary action is governed by policy and procedures applicable to each particular Respondent – staff (HR policies), faculty (HR policies, Board policies, and the Faculty Handbook), or students (student handbook and disciplinary processes).
Mitigating and Aggravating Circumstances
In determining appropriate Administrative Action (remedial in nature) or in recommending Sanctions (disciplinary in nature), the University, at the IDO’s discretion, may consider information deemed relevant by the IDO, including (without limitation) whether the:
- Respondent’s actions were Knowing or Intentional, as opposed to Reckless;
- Research Misconduct was an isolated event or part of a pattern of improper conduct;
- Research Misconduct had a significant impact on the proposed or reported Research Record, subjects, other researchers, institutions, or the public health or welfare;
- Respondent accepted responsibility for the Research Misconduct by: (1) admitting the conduct; (2) cooperating with the Research Misconduct Proceeding; (3) demonstrating remorse and awareness of the significance and seriousness of the Research Misconduct; or (4) taking steps to correct or prevent the recurrence of the Research Misconduct;
- Respondent blamed others rather than accepting responsibility for the questioned conduct;
- Respondent retaliated (or attempted to retaliate) against any Complainant, witness, committee member, or others; and
- Respondent is presently responsible to conduct Research (e.g., has not been debarred from federally-sponsored research), including externally supported Research.
Implementing Administrative Actions
Upon finalizing his/her review of the Investigation report, the IDO may impose Administrative Actions including (without limitation):
- Action taken to preserve and protect human or animal subjects, the safety of personnel, the Research Record, equipment, data, or other property owned by the University or entrusted to its control;
- Required training (or additional training) in the responsible conduct of research;
- Reassignment or removal of personnel in connection with any funded project (including a PI or other investigator);
- Monitoring of the Research process and the handling of funds and equipment;
- Enhanced review or regular audits of IRB or IACUC protocols and compliance;
- Withdrawal, correction, or retraction of pending or published abstracts or manuscripts (may be required as a condition for continued employment);
- Correction or retraction of published materials, including working with journal editors and co-authors to ensure that the Research Record is corrected;
- Internal review of proposals and manuscripts prior to external submission;
- Withdrawal or ongoing monitoring of Research proposals;
- Monitoring of ongoing Research and reporting;
- Required use of plagiarism-detecting software; or
- Delayed or limited approval or submission of manuscripts, publications, funding proposals, or reports.
The Respondent may appeal Administrative Actions through normal appeal procedures, but Administrative Actions will not be held in abeyance pending conclusion of the appeal.
Performance Evaluation Following a Finding of Research Misconduct
If a member of the University staff or faculty is found to have committed Research Misconduct, the employee’s performance evaluation should reflect that finding in enough detail to permit later review by administrators who conduct or review performance evaluations for that employee. Depending on the nature and severity of the Research Misconduct, an overall negative performance rating may result, and corresponding salary implications may follow (e.g., loss of eligibility for merit pay or ineligibility for an across-the-board salary increase). A finding of Research Misconduct should also be considered in tenure and promotion decisions.
Disciplinary Sanctions
Sanctions in response to Research Misconduct may include a range of actions, up to and including termination of employment, pursuant to the procedures set out in the applicable Board policies and faculty handbooks. Sanctions specifically appropriate to cases of Research Misconduct may include (without limitation) one or more of the following:
- Suspension or termination of graduate teaching faculty credentials;
- Bar from recruiting or hiring graduate students or post-doctoral associates;
- Bar from submitting proposals for internal University grants or other competitive funding (e.g., industry grants);
- Limits on access to (or spending of) funds allocated to the faculty member’s discretionary use (e.g., finance and administrative funds or travel funds);
- Limits on the types of Research in which a researcher may engage (e.g., involving human subjects);
- Suspension of employment, with or without pay, for a defined period of time;
- Termination of employment.
Debarment from participation in federal-wide research support (e.g., by PHS or other agency) meets the standard of “Adequate Cause” for termination or other Sanction pursuant to Board Policies Governing Academic Freedom, Responsibility, and Tenure. In any case of federal debarment, the Chief Research Officer will review the circumstances and consider whether Sanctions, up to and including termination of employment, should be imposed.
A Respondent may appeal any Sanction in any manner permitted based on the nature of the Respondent’s employment/affiliation with the University. For example, faculty appeals may be taken through the process described in Board policies and the applicable faculty handbook; students may appeal in a manner described in the applicable student catalog or handbook; and staff may pursue any appeal available under HR policies.
Sanctions against faculty members will be held in abeyance while an appeal is pending only if abeyance is required by Board policy.
Preventing or Mitigating Retaliation
Even after a Research Misconduct Proceeding is concluded, any concern about possible Retaliation against a Complainant or participant in a Research Misconduct Proceeding should be immediately reported to the RIO. The RIO will review the matter with University Counsel and, as necessary, make all reasonable and practical efforts to counter any actual (or potential) Retaliation and to protect and restore the position and reputation of the person against whom the Retaliation has been directed.
Closure of Research Misconduct Proceedings
After consultation with the Chief Research Officer, University Counsel, and federal oversight officials where appropriate, the RIO will provide Notice (to Respondents, Complainants, witnesses, University administrators, and committee members) explaining that the Research Misconduct Proceeding is closed and providing any necessary instructions regarding the RIO’s collection and retention of records.
In cases where an agency’s oversight review is likely to continue, the RIO cannot close the case but may collect and retain records, explaining the impact of ongoing agency review. The University’s Research Misconduct Proceedings may be closed by the RIO after the IDO’s final decision; however, the University remains obligated to assist ORI or other oversight officials in any further review. The University’s proceedings may be reopened at the request of oversight officials.
Measure to Protect or Restore Respondent’s Reputation
The University will undertake all reasonable and practical efforts (if requested, and as appropriate) to protect or restore the reputation of any Respondent against whom no finding of Research Misconduct is made. The Respondent should consult with the RIO to identify and discuss possible restorative measures.
Ongoing Compliance with Administrative Actions, Debarment, or Supervised Research
PHS regulations require that (upon request) the University assist ORI in implementing Administrative Actions imposed by PHS, either during a Research Misconduct Proceeding or after a proceeding is concluded, including (without limitation): debarment from receiving federal grant and contract funds; certification of sources; certification of data; supervision of Research; retraction or modification of articles. The RIO should monitor retractions and modifications by tracking the articles in question for at least one year and should notify editors if modified or retracted publications appear without proper notice of modification or retraction.
Appendix E: Sharing, Retention, and Ownership of Research Data
Research Data are a valuable asset to the University. This policy protects the property rights of individual researchers and the University by addressing definition, responsibility, control, and distribution of certain data. This policy is applicable to Research Data developed by University employees in performing the duties of their University employment or through substantial use of funds and facilities provided by the University. This policy assures that Research Data are adequately recorded, archived, retained, and accessible for sufficient time to support the associated Research that produced the data and any intellectual property based on that Research.
Definition of Research Data
For purposes of this policy, Research Data includes all records necessary for the reconstruction and evaluation of reported results of Research and the events and processes leading to those results, regardless of form or media. Research Data may include laboratory notebooks, databases documenting Research, and other compilations of information developed during Research. Research Data are distinct from, but may be associated with, other intellectual property such as patentable or copyrightable works, and trademarks, which are addressed by other University policies.
Control of Research Data
The University supports the principle of openness in Research. Free dissemination of data, processes, and results of Research and other sponsored activity is crucial to a vibrant and healthy academic environment. The University promotes the prompt and open exchange of Research Data with scientific colleagues outside the investigator’s immediate laboratory or department, subject to relevant grants, contracts or other binding agreements, compliance requirements, or the protection of intellectual property.
In the case of externally sponsored Research involving a grant, contract, or other agreement, the Principal Investigator (PI) is responsible for controlling storage, use, and distribution of Research Data arising from the Research activity, subject to provisions of the applicable grant, contract, or other agreement, University policy, or applicable law. Data generated at the University generally is owned by the University.
If a PI leaves the University and a Research project is to accompany the PI to a new institution, ownership of the Research Data may be transferred with the approval of the Chief Research Officer and with written agreement from the PI’s new institution that ensures clarity regarding in handling:
- custodial and other responsibilities for the Research Data;
- access to the Research Data by the University and other sponsors; and
- protection of the rights of human and animal subjects.
Questions of Research Data ownership or other matters pertaining to the Research Data policy will be resolved by the Chief Research Officer in conformance with University policies. When necessary to assure access to Research Data, the University has the option to take custody of the data in a manner specified by the Chief Research Officer.
University Responsibility for Research Data
The University is ultimately responsible for the accuracy and sufficiency of Research Records, the cornerstone of rigorous Research. Therefore, the University is responsible for Research Data developed by personnel while performing their assigned duties or through substantial use of facilities or funds provided by the University. Such responsibility applies to Research funded by external sources and managed by the University, unless the University agrees to another arrangement in a grant, contract, or other agreement. Specifically, University obligations regarding Research Data include (without limitation):
- Ensuring the academic freedom of the faculty in pursuit of the University’s mission of developing and disseminating new knowledge;
- Securing and protecting intellectual property rights connected with Research Data and commercialization of those rights where appropriate and feasible;
- Protecting the right of access to Research Data, of faculty, postdoctoral scholars, students, and staff;
- Avoiding undue interference with appropriate dissemination of Research Data in the academic community;
- Complying with the terms of sponsored grants, contracts, other agreements, or applicable law;
- Reviewing all charges of Research Misconduct, Conflict of Interest, or similar charges or disputes; and
- Ensuring the appropriate care of animals, human subjects, recombinant DNA, radioactive materials, controlled substances and the like.
Responsibilities of the PI or Laboratory/Department Head Regarding Research Data
Final responsibility and control of Research Data remains at all times subject to the other provisions of this policy. Specifically, however, the PI or the laboratory/department head is responsible for the following:
- Collection of Research Data, including production of defensible laboratory notebooks;
- Management of Research Data to ensure efficient and effective retrieval by the PI, other personnel within project team, or appropriate administrative personnel or research sponsors;
- Development of a formal Research Data plan and procedures where appropriate;
- Consideration of a system for preserving Research Data in the event of a natural disaster or other emergency;
- Retention of Research Data for the requisite period of time (see below);and
- Documented communication of the management system and description of the data managed to members of a research group and to the Chief Research Officer.
Specific Obligations Regarding Preservation of Research Data
The PI or the laboratory/department head must preserve Research Data for a minimum of six (6) years after the final project close-out, with original data retained where feasible, and unless otherwise required by law. The following circumstances may require longer retention:
- If data supports a patent (or patent application), such data must be retained as long as the patent and any derivative patents are valid;
- If Allegations of scientific Research Misconduct, conflict of interest, or other charges arise, data must be retained until such charges are fully resolved plus seven (7) years;
- If a student participates in the project, Research Data must be retained at least until the degree is awarded or the student has unambiguously abandoned the work; and
- As required by the terms of a grant, contract, other agreement, or applicable law.
Beyond these periods, destruction of the Research Record is at the discretion of the PI or the laboratory/department head. Research Data will normally be retained in the administrative unit where generated. Research Data must be retained in a University facility unless specific permission to do otherwise is granted by the Chief Research Officer.
Appendix F: Summary of Protections and Obligations Applicable to the Respondent*
* This appendix includes a summary of specific procedural protections and obligations related to the Respondent but does not replace or alter the specific requirements of the policy or applicable regulations.
During the course of a Research Misconduct Proceeding, the University will make all practical efforts to ensure that core procedural protections are afforded to the Respondent, consistent with obligations to funding agencies and other sponsors.
The Respondent has the right to receive Notice of an Allegation of Research Misconduct and an opportunity to respond to the Allegation. Coincident with those rights are the responsibilities of the Respondent, which are included in the following summary.
Specifically, the University will:
- Make a good faith effort to provide written Notice of the Allegations to the Respondent at the time of, or before, beginning an Inquiry;
- After Sequestration, except as may be deemed necessary by the RIO, provide the Respondent with copies of, or reasonable supervised access to, the Research Record;
- Provide the Respondent an opportunity to comment on the Inquiry report and draft Investigation report and attach to the final report any written comments from the Respondent received within the comment period;
- Notify the Respondent of the outcome of the Inquiry and provide a copy of the final Inquiry report with attachments;
- Provide the Respondent with written Notice of any revised or new Allegations as they may be identified by the RIO, Inquiry committee, or Investigation committee;
- Provide the Respondent a full opportunity to respond to the Allegation in writing, in an interview with the Inquiry or Investigation committee, or both (as the Respondent may choose);
NOTE: While the policy does not require that the Respondent be interviewed during an Inquiry, the Respondent may request an opportunity to respond to the Allegations in an interview with the Inquiry committee, and a timely request to do so will normally be granted, in the RIO’s discretion. Any interview with the Respondent will be recorded or transcribed.
- Interview any witness who has reasonably been identified by the Respondent (during the Investigation) as having information on relevant aspects of the Investigation. Interviews with witnesses will normally be recorded or transcribed;
- Permit the Respondent, at his/her own expense and initiative, to retain and consult with legal counsel or other adviser (who is not also a witness in the proceedings). Respondent’s legal counsel or other adviser may attend the Respondent’s interview with an Inquiry or Investigation committee and provide advice to the Respondent during the interview, so long as the legal counsel or adviser does not address the committee directly or otherwise interfere with the proceedings.
- When no finding of Research Misconduct is made, undertake all reasonable and practical efforts to protect or restore the reputation of such a Respondent, if requested and as appropriate, and in consultation with the Respondent;
- Provide an opportunity for the Respondent to appeal – pursuant to the applicable appeal procedures – any Administrative Actions or Sanctions imposed as a result of a finding or admission of Research Misconduct.
During a Research Misconduct Proceeding, the Respondent must continue to comply with all applicable University rules and policies. The University expects that the Respondent will:
- Conduct him/herself professionally and in accord with specific instructions of the RIO, IDO, Chief Research Officer, or other University official implementing this policy;
- Cooperate fully in the Research Misconduct Proceeding;
- Provide the RIO with all requested information or evidence, including relevant aspects of the Research Record of which the RIO may not be aware, and assist the RIO in locating any evidence that may be relevant but not within the Respondent’s immediate control; and
- Refrain from any form of Retaliation against any Complainant known or suspected by the Respondent. Retaliation for making a Good Faith Allegation or for Good Faith participation in a Research Misconduct Proceeding is specifically prohibited by federal law and by this policy.
- The Respondent should take care to avoid even the appearance of Retaliation against a person making an Allegation or cooperating with the Research Misconduct Proceeding. If the Respondent has a question about whether contemplated action might constitute prohibited Retaliation, he/she should seek the advice of the RIO.
- If a Respondent believes that an Allegation has been made in bad faith, he/she should provide a written explanation of that concern to the RIO, who will follow this policy with respect to an Allegation made in bad faith. Raising a concern of bad faith does not entitle the Respondent to suspension or modification of ongoing Research Misconduct Proceedings.
Respondent Acknowledgement receipt of Summary of Date
Protections and Obligations Applicable to the Respondent
Research Integrity Officer Date
Appendix G: Sample Allegation Intake Form
|
Date | |
|
Case ID | |
|
Research Integrity Officer (RIO) | |
|
Complainant (contact information and relationship to possible Respondents or witnesses) | |
|
Possible Respondent(s) (title and contact information) | |
|
Other collaborators, co-authors, or witnesses, at UT or elsewhere | |
|
Possible funding sources | |
|
Human or animal subjects | |
|
Hazardous materials or biologics | |
|
ALLEGATION(S) (identify available detail about specific Falsification, Fabrication, or Plagiarism alleged | |
|
Possible evidence in support (or rebuttal) of Allegation(s) | |
|
Location and nature of possible evidence requiring Sequestration | |
|
Concerns and warnings against Retaliation | |
|
RIO notes (including any concern about whether the Allegation was not made in Good Faith) |






